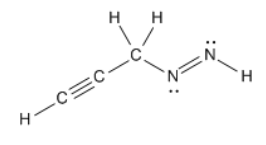
When two s-orbitals of the opposite sign interact, they form a `sigma"-antibonding"` orbital. When two s-orbitals of the same sign interact, they form a `sigma"-bonding"` orbital. In MO theory, when two orbitals interact, they form a set of molecular orbitals. 
The results of these two are shown in the images below: What happens when s-orbitals interact? There are two possibilities: the first of which being that two s-orbitals of the same sign interact, and the second of which being that s-orbitals of the opposite sign interact. 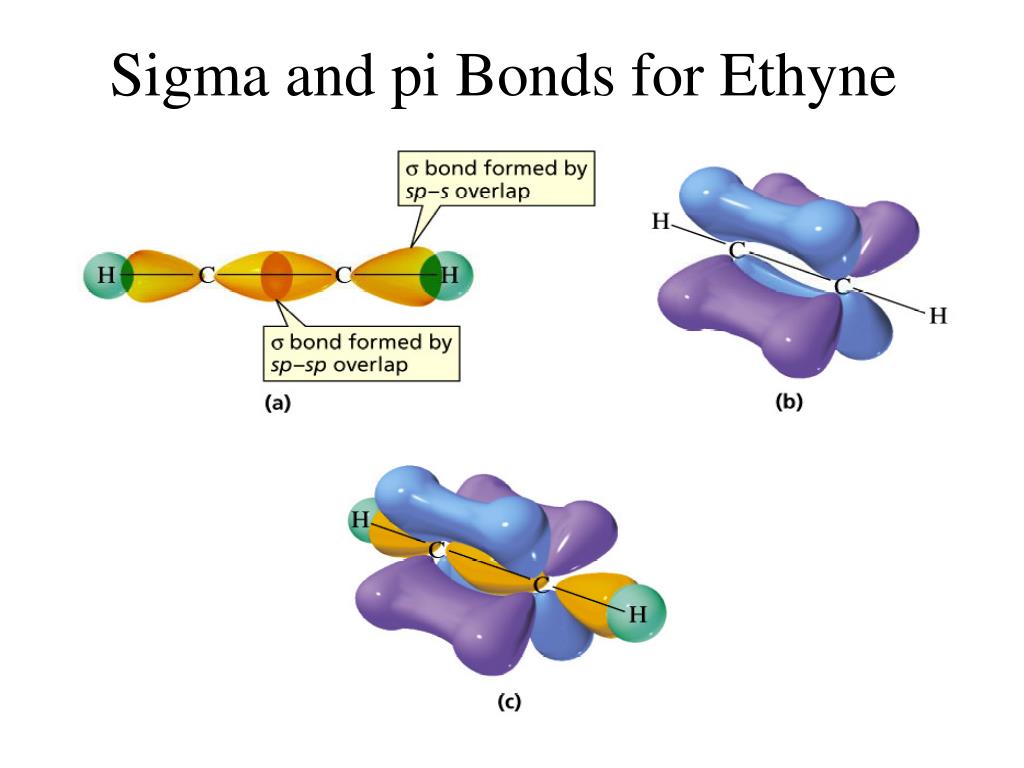
S-orbitals are usually written as having one of two colors. We'll call this the "sign" of the orbital. These states are often written as `+` and `-`, or drawn as different colors.įor example, the p-orbitals are usually drawn with the two regions as different colors. It turns out that, according to the wave equation, orbitals can be one of two states. Unfortunately, MO Theory can be the most difficult to understand and visualize, which is why we covered the other two first.Īs we mentioned in earlier posts, the way we determined the shape of orbitals is through Schrodinger's wave equation. The purpose of MO theory is to fill in the gap for some behavior that cannot be explained by VSEPR and Valence-Bond Theory. In contrast to VSEPR and valence bond theory which describe bonding in terms of atomic orbitals, molecular orbital theory visualizes bonding in relation to molecular orbitals, which are orbitals that surround the entire molecule. Molecular Orbital (MO) Theory is the final theory pertaining to the bonding between molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
